Intracellular membrane trafficking in the nervous and neuroendocrine systems
Stéphane GASMAN & Nicolas VITALE

 ⇨ Teams
⇨ Teams Presentation
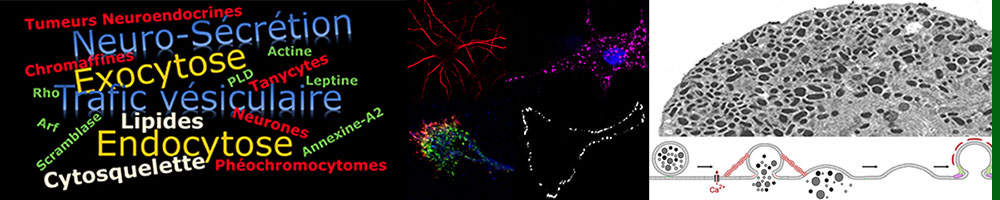
Functioning of the nervous and neuroendocrine systems is based on the release of informative molecules thanks to various tightly regulated membrane trafficking processes (exocytosis, endocytosis, vesicular transport). 
Our research aims at deciphering the molecular mechanisms regulating these trafficking events and at unravelling their dysfunction associated with important pathologies (mental retardation, neurodegenerative diseases, neuroendocrine cancers, obesity…).
Using neurons, neuroendocrine cells and glial cells, we are particularly studying the mechanisms by which different lipids and proteins control the life cycle of vesicles, starting from biogenesis to membrane fusion through their intracellular transport.
> Formation of exocytosis sites
Sylvette CHASSEROT-GOLAZ
> Membrane contact sites and regulated exocytosis
Sylvette CHASSEROT-GOLAZ, Nicolas VITALE
> Neuroendocrine secretions and tumors
Stéphane GASMAN
> Transport of metabolic hormones to the brain
Stéphane ORY, Stéphane GASMAN
> Copper and amylin in Alzheimer disease
Nicolas VITALE

(Ph.D. student)
The molecular basis of hormone secretion and neurotransmission has long been investigated through the lens of protein machineries responsible for vesicle fusion and recycling. However, the contribution of membrane lipids to these processes remains largely underestimated. Plasma membrane phospholipids are asymmetrically distributed between the two leaflets of the lipid bilayer, conferring a fundamental structural and functional polarity to the membrane. Our work has shown that a local and transient loss of this asymmetry is not merely a consequence, but a prerequisite for hormone secretion in neuroendocrine cells and for neurotransmission at high-frequency synapses. We have identified Scramblase-1 (PLSCR1) as a central regulator of this process. This protein locally perturbs lipid asymmetry near vesicle fusion sites and regulates vesicle recycling through compensatory endocytosis. My research aims to decipher how PLSCR1 orchestrates the coupling between exocytosis and endocytosis, and to establish how dynamic changes in membrane asymmetry govern the exo-endocytic cycle of secretory vesicles. To address these questions, we combine advanced cell biology approaches with high-resolution imaging techniques in primary cultures of cerebellar granule neurons.

(Ph. D. student)
My thesis project on Alzheimer’s disease and type 2 diabetes lies at the interface between chemistry and biology and is funded by the CNRS MITI 80′ program. Both diseases involve excessive production of peptides capable of self-assembly into fibers which are associated with a certain degree of cellular toxicity. These are amyloid-beta peptides in Alzheimer’s and amylin in type 2 diabetes. By carrying out co-assembly kinetics studies on the formation of co-aggregates, I’m investigating the impact that these two peptides can have on each other. In parallel, structural studies will enable us to observe and compare the fibers formed. The impact of these co-aggregates on organotypic cultures of mouse hippocampus is evaluated to study their toxicity by immunocytochemical approaches and by measuring neuronal network activity by MEA. This thesis aims to establish whether amylin does indeed act as a link between these two pathologies and is co-directed by the Laboratoire de Chimie de Coordination in Toulouse and INCI in Strasbourg.

(Post-doc)
The role of lipids, major components of cellular membranes, in the release of neurotransmitters and hormones is still poorly understood, mainly because of a lack of efficient tools. My work is based on the usage of optogenetic tools and ‘click chemistry’ to, on one hand, direct lipid metabolism enzymes to a specific membrane compartment and, on the other hand, introduce ‘clickable’ lipids into membranes allowing the grafting of various functional groups. By employing these innovative tools on bovine chromaffin cells, combined with advanced microscopy approaches or electrochemistry, I explore the pleiotropic roles of lipids in neurosecretion with unprecedented spatial and temporal precision.

(Post-doc)
My postdoctoral project, funded by an ANR contract, aims to better understand the role of membrane contact sites (MCS) in regulating exocytosis, an essential cellular process that enables the release of hormones, proteins, and neurotransmitters. Although the central mechanisms of vesicle fusion are well understood, the fine mechanisms that prevent random secretion remain poorly understood. My goal is to study the formation and dynamics of these MCS, their influence on calcium homeostasis near exocytosis sites, and their role in the transfer of lipids essential for proper secretion. To do this, I will combine advanced imaging, biochemistry, and electrochemistry investigations on cellular and murine models. In this way, I aim to generate new knowledge about intracellular communication and the regulation of a major biological process, with potential implications for understanding certain pathologies linked to exocytosis defects.
Our Publications